Translate this page into:
Modeling one-stop-shop integration of family planning and HIV services in Zimbabwe, 2021

*Corresponding author: Hamufare Dumisani Mugauri, AIDS and TB Unit, Ministry of Health and Childcare, Harare, Zimbabwe. dumiwaboka@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mugauri HD, Chikondowa I, Kanyowa T, Ngwenya M, Macheka M, Murwira M, et al. Modeling one-stop-shop integration of family planning and HIV services in Zimbabwe, 2021. J Reprod Healthc Med 2023;4:6.
Abstract
Objectives:
In October 2020, the Ministry of Health and Child-care Zimbabwe commenced integrating reproductive health with human immunodeficiency virus (HIV) services. Family planning (FP), sexually transmitted infection (STI) screening and treatment, HIV testing services (HTS), pre-exposure prophylaxis (PrEP), and antiretroviral therapy (ART) services were offered through an integrated one-stop-shop model. The project aimed to address unacceptably high incidences of HIV and STIs among FP clients and demonstrate improved access and uptake of integrated services at dedicated FP centers.
Materials and Methods:
We conducted an intervention study. A service package of STI symptom screening and HTS (including HIV self-test [HIVST] primary and secondary distribution) was offered to all FP clients by multi-skilled nurses. Service uptake was measured pre and post-introduction of the intervention, before which services were provided by different nurses, in different rooms. HIV-diagnosed clients were linked to ART and eligible negative testers were offered PrEP. Service uptake was measured at 1 year of implementation against the same period before.
Results:
A total of 20,947 clients were recruited between October 2019 and September 2021, 9386 pre-intervention and 11,561 post-intervention. We observed a 5.2% total increase in clients seen and a 27.1% increase in the average uptake of all integrated services (FP, STI, HTS, PrEP, and ART). Most (93.5%) clients received oral contraception (Progestogen-only pills and combined-oral-contraceptives), while fewer (4.8%) received injectables and implants (1.1%). Following the intervention, STI screening increased by 30.2% (2730 vs. 11,028). HTS delivery increased by 38.9% and PrEP by 44.1%. HIV positivity yield increased significantly (10.1%, 62/615 vs. 16.6%, 813/4910) and linkage to ART improved by 44.4%. HIVST distribution contributed to improved access to HIV testing with 3754 FP clients accepting distribution compared with 4910 receiving HTS by nurses, provider-delivered testing was accessed by reactive self-testers. The project increased (?) improved linkage to preventive and treatment services and reduced missed opportunities associated with fragmented services.
Conclusion:
The integrated one-stop-shop model demonstrated improved service uptake during the intervention period. This project shows the benefits of integrated FP/STI/HTS/PrEP in Zimbabwe’s high HIV burden settings.
Keywords
Integration
One-stop-shop model
Linkage
Sexual and reproductive health services
HIV testing services
Pre-exposure prophylaxis
INTRODUCTION
In parts of East and Southern Africa, approximately 60% of new HIV infections are among women and girls of all ages.[1] In sub-Saharan Africa, gender inequality contributes to the heightened risk of HIV faced by adolescent girls and young women in the region: Six in seven new HIV infections among adolescents (aged 15–19 years) in the region occur among girls, and young women (aged 15– 24 years) are twice as likely to be living with HIV as young men of the same age.[2]
At the same time, in Southern Africa, nearly one in every four women ages 15–19, and nearly one in every five aged 15–49, are preoccupied with avoiding pregnancy without considering their risk of contracting HIV.[3] Located in Southern Africa, HIV prevalence in Zimbabwe is estimated at 12.9% and is largely driven by unprotected sexual intercourse.[4]
Despite the evidence and opportunities, implementation of HIV testing in family planning (FP) in Zimbabwe remains limited due to systems barriers, such as different cadres, workflow, funding, monitoring, and reporting, as well as fears of stigma and discrimination.[5] To reach women and their partners who are most affected by and at risk of HIV acquisition, greater efforts to integrate services are needed to achieve impact.[6] In addition, COVID-19 has affected and disrupted the delivery of health services including HIV testing and prevention, and FP, particularly in facilities.[7] Introducing feasible ways to maintain and expand services, particularly through HIV self-testing (HIVST), during this time are critical.
Concrete action is required, particularly in Zimbabwe, to expand coverage of services for FP/contraception and HIV testing services (HTS) and linkage to HIV prevention and treatment for women and adolescent girls. In settings, such as Zimbabwe, where the HIV burden is high, FP clients should be routinely offered HIV testing as well as a choice of contraceptive methods.[5,8] Integrated FP services and HTS can be implemented through a variety of provider-delivered and self-testing modalities, such as offering HIVST in FP services provided in health facilities, pharmacies, and communities.[9] HIV self-tests can be distributed to clients at facilities (facility-based distribution) and within the communities (community-based distribution). Service delivery methods include primary distribution where an individual obtains a self-test from a provider or secondary distribution whereby a self-test -kit is collected on behalf of the person who will test, mostly a partner.
In May 2020, the Government of Zimbabwe embarked on a pilot project to integrate FP with sexually transmitted infection (STI), and HIV testing, including HIVST, and pre-exposure prophylaxis (PrEP) services. The approach was informed by the evidence of contraceptive options and HIV outcomes trials which showed that settings with high background HIV prevalence and incidence had unacceptably high rates of HIV and STI infections among women attending FP clinics. Programmatic data in Zimbabwe indicated that most (94%) women of reproductive health prefer non-barrier methods of FP leaving them exposed to the risk of HIV infection through unprotected sexual intercourse.[10] Further, these women experience low levels of HIV testing because they access FP services mostly through dedicated FP services where reproductive and HIV service provision is not synchronized to provide a comprehensive package. The study was piloted in four FP clinics managed by Zimbabwe National Family Planning Council (ZNFPC) in Harare (Spilhaus and Fife Avenue) and Bulawayo (Lister and Mpilo Centre).
In this new “one-stop-shop” model, trained FP providers deliver an improved minimum package of FP, STI, and HTS services (including primary and secondary distribution of HIVST) for all clients in contrast with the previous supermarket approach, as shown in [Figure 1]. All clients presenting for FP services were offered a package of services that consisted of STI screening, and HIV testing (including HIVST and secondary distribution for partners). Following the HIV test outcome, subsequent services were offered; clients with HIV negative test results are offered PrEP depending on the client’s HIV risk profile; clients who test positive are linked for initiation on antiretroviral therapy (ART). Partner testing and voluntary-assisted partner notification appropriate to test results are offered to FP clients who receive facility-based testing. HIVST information and kits are offered to women during post-test counseling. To facilitate acceptance of the HIVST by the partner, and to promote disclosure where appropriate, two test kits are offered to each FP client.

- Contrasting the Status quo (Supermarket approach) with the one-stop-shop model of integration, Zimbabwe, 2020.
To facilitate the implementation of this project, the Government of Zimbabwe with support from its implementation partners had to propose the pilot with ZNFPC leadership to obtain buy-in to change their service delivery protocol. Other requirements for the success of the project included capacity building of staff to be multi-skilled and provide a comprehensive package within the same service delivery space and developing indicator tracking mechanisms for perioding evaluation of performance.
The main objective of this study, conducted in Harare and Bulawayo, Zimbabwe was to demonstrate improved service uptake when services are packaged into a “One-stop-shop model” of integration. To achieve this, the study aimed to (i) describe the demographic data of clients over the study period, (ii) measure the uptake of the services pre- and post-intervention, and (iii) describe the monthly trends for services uptake between April 2020 and March 2021.
MATERIAL AND METHODS
Study design
This was an implementation study with a pre-and post-intervention phase.
Setting
General setting
Zimbabwe is a landlocked, low-income country in Southern Africa located between Botswana, South Africa, Mozambique, and Zambia with an estimated population of 16 million people.[11] In 2020, Zimbabwe had an estimated population of 16 million and ranked number 154 out of 189 countries on the human development index.[11,12] In the same year, Harare and Bulawayo, the two largest cities recorded an HIV prevalence of 12.2% and 14%, respectively, compared with the national average of 12.9%.[4]
The AIDS and TB program (ATP) coordinates the development of national HIV health policies and establishes national standards and guidelines as part of the national response to HIV in Zimbabwe. Four sub-units under ATP, namely, HIV Prevention, Care and Treatment, prevention of mother-to-child transmission (PMTCT), and Monitoring and Evaluation, are mandated to ensure seamless yet specialized programming to ensure adequate response to the pandemic.[13] ZNFPC, on the other end, is a parastatal organization that runs a total of 11 dedicated FP clinics, dotted around the country.
Study sites
Harare is the capital city of Zimbabwe with an estimated population of 2 million in 2017.[14] It has a total of 36 clinics of which 12 are polyclinics and an additional two hospitals run by the local authority.[15] The city also hosts two quaternary health centers that serve as the largest referral centers in the country. The city also houses two dedicated FP centers, namely, Spilhaus, which also serves as the headquarters for the organization, and Fife Avenue which mainly caters to clients on the Western side of the city. Spilhaus was offering HTS, STI, and PreP services in addition to FP pre-intervention utilizing the supermarket approach before the intervention.
Bulawayo is Zimbabwe’s second-largest city and had a population of 0.74 million in 2017.[14] It has a total of 19 primary health-care clinics, one laboratory and one infectious disease hospital under the municipality.[16] Bulawayo houses Lister House, a dedicated FP center for Bulawayo city. A second facility, Mpilo (also referred to as Matabeleland North), is housed within Mpilo Central Hospital’s yard.
Patient population
All clients, 16 years and above, who reported for FP services at Spilhaus, Fife Avenue, Lister house, and Mpilo service centers from April 1, 2020, to March 31, 2021, and were documented in the FP register were included in this study. The same clients were tracked for uptake of HTS, HIVST, STI, and PrEP services as documented in the program as well as summary registers.
Data variables, sources of data, and data collection
Data were collected between April 2020 and March 2021 using a structured data collection register that tracked summary indicators of all the integrated services. A line list of diagnosed patients was prepared from the laboratory register after the removal of duplicate entries.
The following variables were collected: Name of the patient, age, sex, FP choice, screened for STI (yes/no), HIV status, tested for HIV (yes/no) and result (positive/negative), date of testing, HIVST provided for primary and secondary distribution (yes/no), eligible for PrEP (yes/no), and initiated or linked for PrEP which was not available, linked for ART (yes/no/NA). The sources of data included the FP register, HTS register, FP register, HIVST register, PrEP register, and integration register. If the patient was not documented to be registered in the integration register, we confirmed the reception of integrated services in the respective individual registers. Linkage to service was considered for clients who received the referred services.
Analysis and statistics
Data were collated onto an Excel sheet [S1 Annex] and read through EpiData (version 3.1 for entry and version 2.2.2.183 for analysis, EpiData Association, Odense, Denmark) for descriptive analysis. The trends of service uptake 1-year pre-intervention and 1-year months post-intervention, between April 2020 and March 2021 were summarized. Key outputs were the number and proportion of clients receiving services in the pre-and post-intervention phases, namely, FP, STI, HTS, PrEP, and ART.
Ethics approval
This study was approved by the Ministry of Health and Child Care and ZNFPC. The approval took into account that the data utilized are routinely collected for programming and the interviews conducted were part of routine Support and Mentorship activities, which are a component of programming.
RESULTS
Baseline characteristics
A total of 20,947 clients presented for services at the four sites sampled for this study (Spilhaus, Fife Avenue Clinic, Lister House, and Mpilo Central Hospital site) from October 1, 2019, to September 31, 2021, divided into 1-year pre-intervention (9386) and 1 year post the intervention (11,561). The average increase in service uptake for the integrated services (FP, STI, HTS, PrEP, and ART) was 27.1% comparing the pre and post-intervention periods.
In the post-intervention phase, a 5.2% increase in the total number of clients who reported for FP services was observed. Most clients were between 15 and 24 years (32.5%, n = 6818) both pre- and post-intervention, translating to a 9.3% increase in the post-intervention phase. This was followed by the 45–54 year category which gained 6.6% in the post-intervention phase (n = 1239 vs. 1619). The highest number of clients were seen at Lister House (8738) with 50.8 % of these being seen in the post-intervention phase. Only Fife Street recorded a decline in the number of clients seen in the post-intervention phase, constituting 47.9% of the total clients seen at this facility. All integrated services recorded an increased uptake in the post-intervention phase, with the highest being linkage to ART at 94.4, 3%, while linkage to PrEP came closely second at 94.1, 1%.
A significant increase in STI screening was observed, 2730 client visits pre-intervention, and rose to 11,028 (80.2%) post-intervention. HIVST was introduced as part of the intervention, with primary and secondary distribution for partners offered to complement provider-delivered HIV testing. Following the intervention, 92.9% (813) of clients tested for HIV turned out positive, translating to a positivity yield rate of 16.6%. The positivity included clients identified through HIVST who scored a reactivity rate of 4.2% (813/3754) [Table 1].
| Variable | Pre- intervention* |
Post- intervention** |
%*** |
|---|---|---|---|
| Total | 9386 | 11,561 | 55.2 |
| Age in years | |||
| 15–24 | 2774 | 4044 | 59.3 |
| 25–34 | 2297 | 2455 | 51.7 |
| 35–44 | 1759 | 1998 | 53.2 |
| 45–54 | 1239 | 1619 | 56.6 |
| 55–64 | 982 | 1098 | 52.8 |
| ≥65 | 324 | 339 | 51.1 |
| Unknown | 11 | 8 | 42.1 |
| Facility | |||
| Spilhaus | 1,957 | 3292 | 62.7 |
| Fife street | 2121 | 1,951 | 47.9 |
| Lister house | 4297 | 4441 | 50.8 |
| Mpilo | 1011 | 1877 | 65.0 |
| Services received | |||
| Family planning | 9386 | 11,561 | 55.2 |
| STI screening | 2730 | 11,028 | 80.2 |
| HIV testing | 615 | 4910 | 88.9 |
| Linked for PrEP | 17 | 269 | 94.1 |
| Linked for ART | 35 | 587 | 94.4 |
| HIVST (Primary) | - | 1968 | 100 |
| HIVST (Secondary) | - | 1786 | 100 |
| HIV result | |||
| Tested positive | 62 | 813 | 92.9 |
| Reactive HVST | - | 157 | 100 |
Access to FP Services
The trends for overall service uptake disaggregated by health facility fluctuated continually with a marginal increase being recorded in the post-intervention phase. Sharp fluctuations were recorded at Lister House, which also recorded the highest number of clients taking up the services over the pre and post-phase of the project. Mpilo recorded no clients in April, September, and October 2020 during which period the site was closed due to COVID-19-induced closure. The other sites recorded fewer clients during the same period [Figure 2].
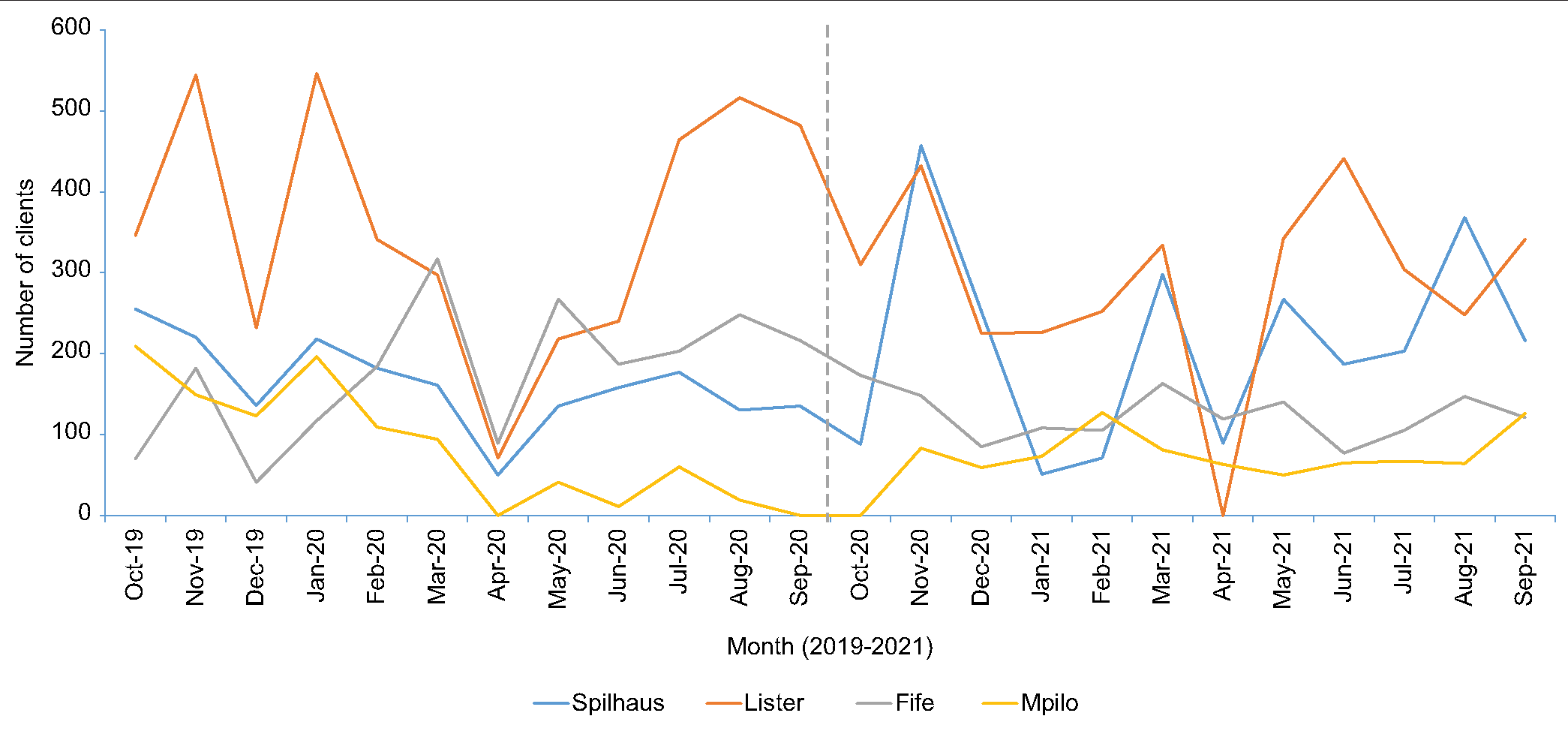
- Trends of clients accessing family planning services by facility, October 2019–September 2021.
Screening for STI
STI screening increased at all centers following the intervention despite a sharp drop being experienced from April 2020 to December 2020. In April, strict lockdown measures were instituted following the COVID-19 outbreak. During this period, all service centers were closed for – 2 weeks [Figure 3]. During the intervention, clients seeking FP services were routinely offered screening for STI at all health facilities and STI screening became the most accessed. STI screening became the most accessed service in the after phase of the intervention [Figure 4].
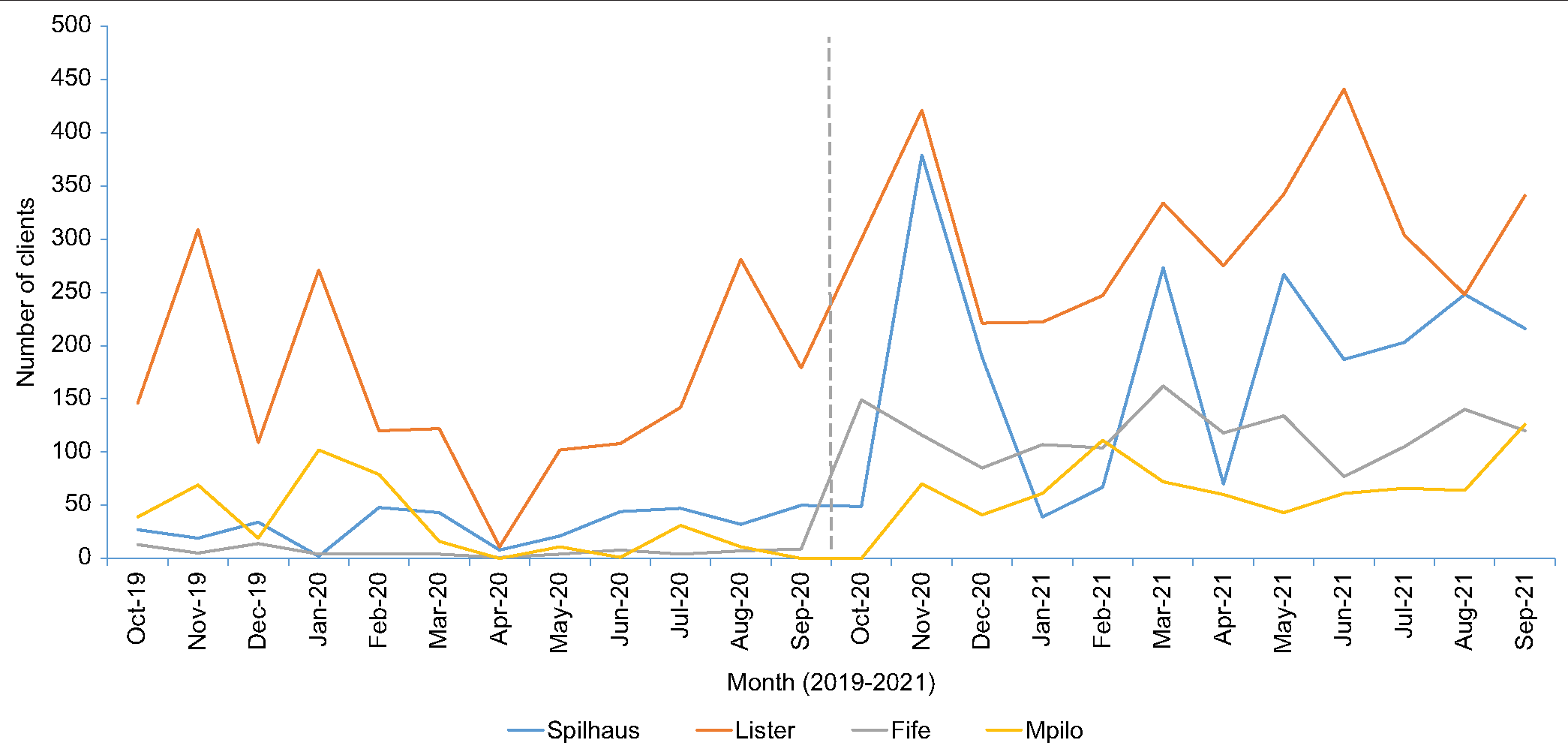
- Trends of clients screened for sexually transmitted infections by health-care facility, October 2019–September 2021.
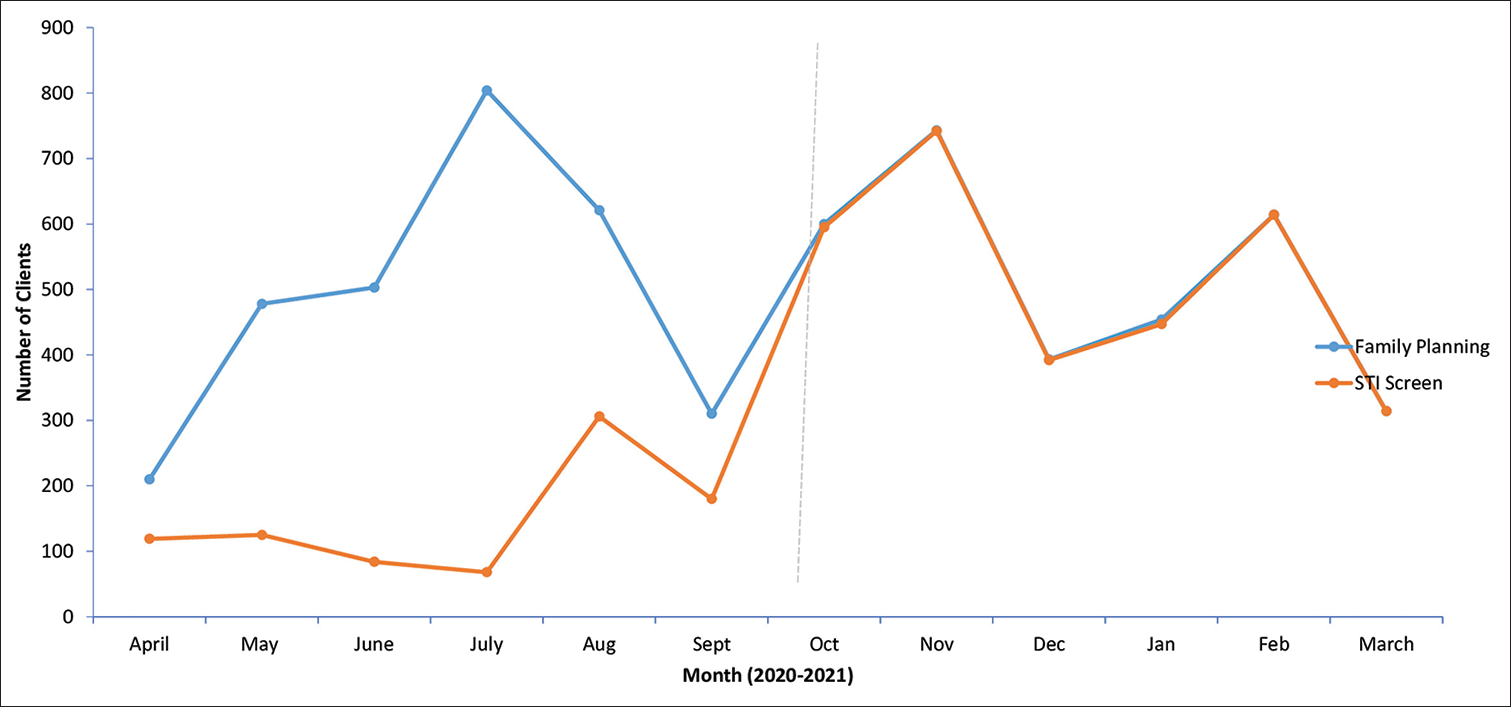
- Trends of family planning clients screened for sexually transmitted infection, October 2019–September 2021.
Testing for HIV (provider-delivered testing) and positivity yield
Provider-delivered HIV testing increased sharply following the intervention in October 2020. Notably, Lister House recorded <60 clients accessing HIV testing throughout the pre-intervention phase, rising to 90 clients being the lowest in the post-intervention phase. This was despite the return of strict COVID-19 lockdown measures being instituted in January 2021, after relief from July 2020 [Figure 5]. HIV test positivity also increased during the intervention, during the pre-intervention phase test positivity was 10.1% (62/615), while, during the intervention, it grew to 16.6% (813/4910) [S. Figure 1].

- Trends of clients tested for human immunodeficiency virus by health-care facility, October 2019–September 2021.
Linkage for PrEP
Among clients seeking FP services, linkage for PrEP services improved overall in the post-intervention phase with the highest point being September 2021 when 34 clients were linked for the service. Stockout of PrEP medicines resulted in a reduction in PrEP provision in December 2020 when only ten clients accessed the service, becoming the lowest point in the after phase. The increase in the clients accessing HIV testing corresponded with the increased number of positives identified with the highest number in the implementation phase being 115 HIV positives in May 2021, in contrast to the highest point of 12 clients in July 2020 during the pre-intervention phase [Figure 6].
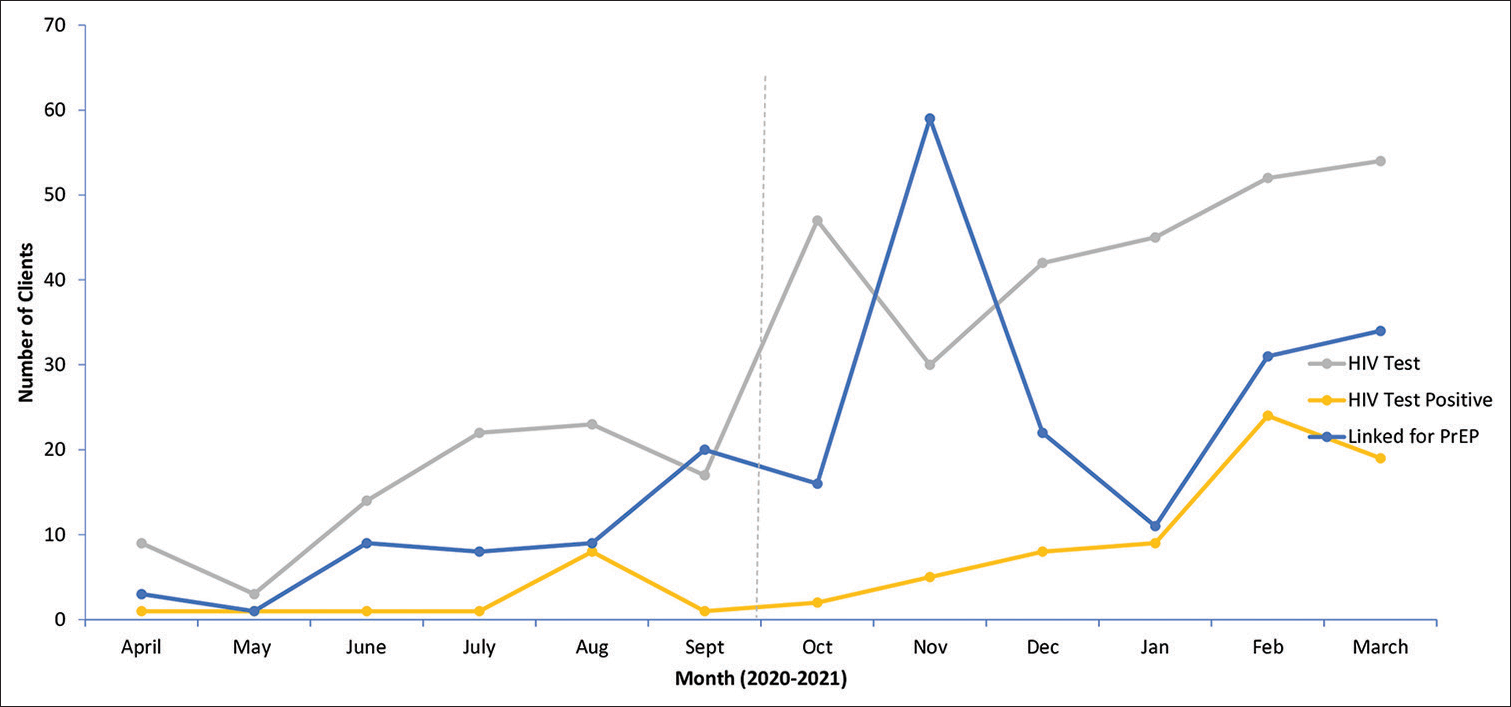
- Trends of family planning clients accessing human immunodeficiency virus services, October 2019–September 2021.
Access to HIVST
IVST, including primary and secondary distribution, was introduced with the integrated service package. The highest uptake of both primary and secondary distribution was in August 2021 at 261 clients and 185, respectively. The uptake of HIVST sharply declined in December 2020 (total = 132) before rising gradually throughout 2021. The reactivity rate has remained low, contributed by the low sharing of results, particularly for secondary distribution. Overall, HIVST improved access to HIV testing resulting in a total of 3754 clients accessing HIV testing through HIVST compared with a total of 4910 clients tested by the provider during the post-intervention phase. Over time, more clients were willing to take an HIVST kit for their partners, as indicated by secondary distribution rising in 2021 [Figure 7].
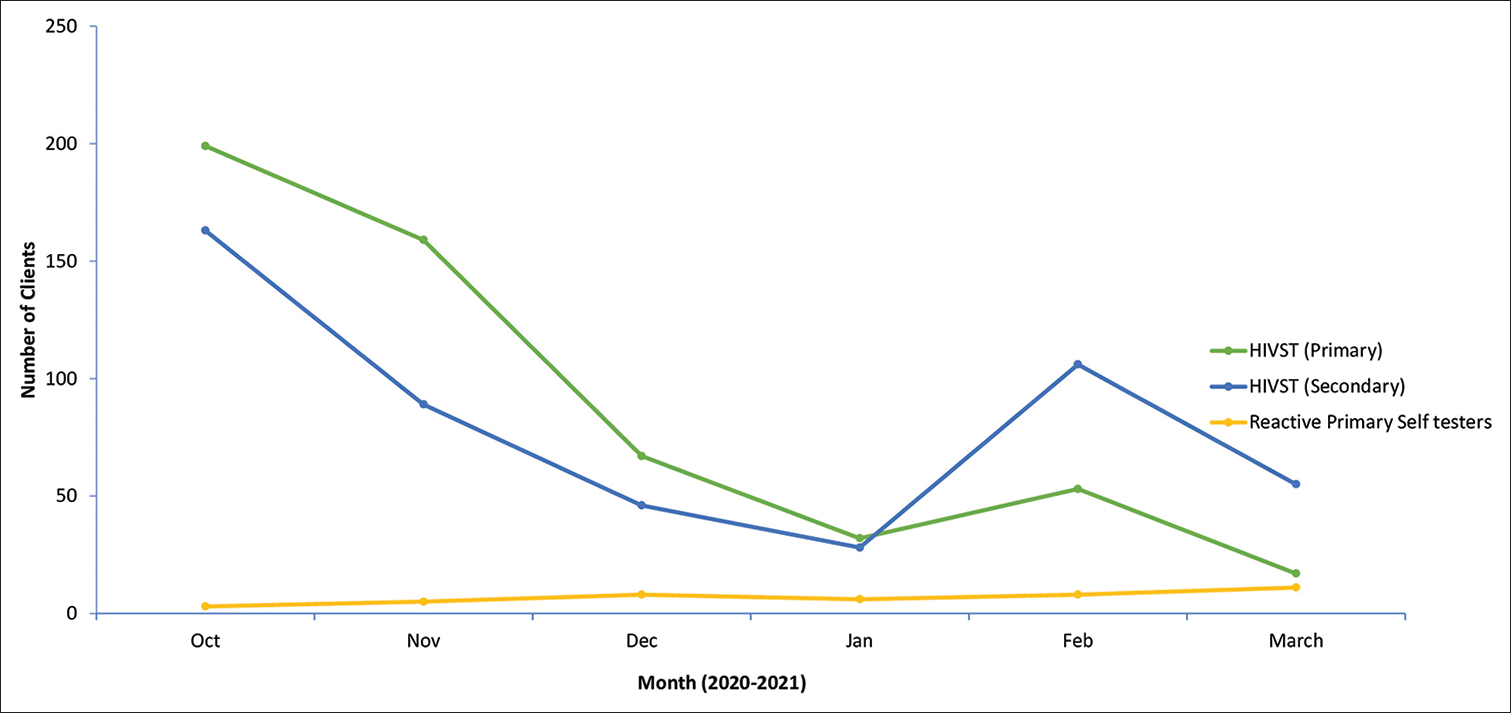
- Trends of family planning clients accessing human immunodeficiency virus self-testing, October 2019–September 2021.
DISCUSSION
Increased access and uptake of integrated services were observed in the post-intervention phase compared with the pre-intervention phase. Most clients accepted integrated services offered to them within the same room and by the same health worker who addressed their FP needs, their primary purpose of visit.
Healthcare workers were capacitated to ensure the implementation of the integrated service package at the commencement of the intervention. Integration indicators were tracked using a summary sheet that was introduced at the commencement of the project to ensure the computation of service uptake of the integrated services and facilitate analysis. The study was done using routinely collected programmatic data which were representative of the reality on the ground.
External linkage of clients, particularly for ART services may have influenced the low uptake of services. This was influenced by the lack of skills to initiate clients on ART by the health workers pre-intervention. However, this was later mitigated by training the health worker on HIV integrated training that encompasses ART initiation and monitoring, in addition to PrEP provision.
Interpretation of key findings
This study provides important insights into the one-stop-shop model of integration at dedicated FP centers.
During the intervention improved, access to the integrated services (STI, HTS, HIVST, and PrEP) was observed. Offering these services within the same room and being offered to clients who presented for FP resulted in clients accepting the additional services. Therefore, the improved uptake may have resulted from easy access to services that were offered within the same room during the intervention, while clients had to move from one room to the next to access the services before it. Similar findings have been documented for SRH services and other services.[17-19]
Second, the one-stop-shop model enabled a person-centered, continuous therapeutic dialogue with the client that connected the client’s choice of FP, risk of HIV and STI acquisition, and pre-test information for HIV testing. All services were linked in a logical flow that appealed to the clients and stimulated their conscience to appreciate their risk for HIV.[20-24] This addressed the preoccupation with birth control which is associated with low HIV risk perception documented in the literature.[25]
Third, high positivity of HIV infection among women seeking FP services was observed. The possible reasons for this were (i) improved access to HIV testing among this group and (ii) a true reflection of higher incidences of HIV infection among women seeking FP. Both assumptions are supported in the literature[19,20,26-28] This highlights the need to improve access to HIV testing through provider-initiated measures rather than waiting for the women to demand the services.
Fourth, in addition to improved access to provider-delivered testing, HIVST promoted access to HIV testing among some women. HIVST also facilitated partner testing, through the secondary distribution of HIVST kits whereby women were able to self-test with their sexual partners, who do not usually accompany them to the clinic. Case finding: Where the women tested HIV negative at the facility, HIVST kits to test with partners facilitated the identification of partners living with HIV. While evidence of the feasibility of this approach is documented in the literature, program examples of this implementation are limited in the FP context.[29-32]
Finally, in this work, we have demonstrated the practice of the facility-based distribution of HIVST in FP services. This testing approach increased positivity yield on provider-delivered HIV testing platforms, demonstrating successful triaging of clients using self-test. This modality promotes autonomy among clients and improved efficiency whereby the health worker concentrates on reactive self-testers to perform confirmatory testing. These benefits of HIVST were observed in the previous studies.[32,33] In this regard, HIVST serves as a screening test to prepare the client for confirmation when a reactive result is obtained.
Implications for policy and practice
Integrating Sexual and Reproductive Health services through the one-stop-shop model demonstrated improved access, uptake, and acceptability of these services highlighting the importance of policy on integration, particularly for dedicated FP clinics.
Dedicated FP centers in Zimbabwe should consider adopting integrated services to deliver person-centered reproductive health services.
Future research
A qualitative study may assist to explore the determinants of varied choices of FP with the view to enhancing the uptake of a variety of short- and long-term methods that are not commonly used in Zimbabwe.
CONCLUSION
The one-stop-shop model integrated (FP, STI, HTS, HIVST, PrEP, and ART) and demonstrated improved service uptake across the range of services. Despite COVID-19-induced social measures that restricted client movement and access to health services.
Authors’ contributions
Conception and design: HDM, IC, TK, MN, OM. Development of data capture tools: HDM, IC, AZ. Data collection: HDM, IC, AZ. Data entry: HDM, IC, AZ. Data analysis and interpretation: All authors. Preparing the first draft of the manuscript: All authors. Critical review and approval of final draft: All authors.
Acknowledgments
We acknowledge the contribution of the Director AIDS and TB unit and the entire team at the Ministry of Health and Child Care, including the Reproductive Health Department who participated in the implementation of this project. We also acknowledge the role of the executive director for ZNFPC and his entire team for their contributions to the success of this project.
Special gratitude goes to the World Health Organization (WHO) head office (Geneva) as well as the Zimbabwe country office for supporting the implementation of this project to its logical fulfillment in generating evidence that supports the one-stop-shop model of integration.
Additional files
Additional file 1: Integration project Data file, excel format for April 2020 to March 2021
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Available from: https://www.kff.org/global-health-policy/fact-sheet/the-global-hivaids-epidemic [Last accessed on 2021 May 26]
- World AIDS Day Report. 2021. Available from: https://www.unaids.org/sites/default/files/media_asset/2021_WAD_report_en.pdf
- [Google Scholar]
- Adolescents, young people, and the 90-90-90 goals: A call to improve HIV testing and linkage to treatment. AIDS. 2017;31(Suppl 3):S191-4.
- [CrossRef] [PubMed] [Google Scholar]
- Integration of HIV-sexual reproductive health services for young people and the barriers at public health facilities in Mbarara municipality, Southwestern Uganda: A qualitative assessment. Int J Reprod Med. 2019;2019:6725432.
- [CrossRef] [PubMed] [Google Scholar]
- Addressing critical gaps in achieving universal access to sexual and reproductive health (SRH): The case for improving adolescent SRH, preventing unsafe abortion, and enhancing linkages between SRH and HIV interventions. Int J Gynaecol Obstet. 2010;110(Suppl):S3-6.
- [CrossRef] [PubMed] [Google Scholar]
- Ramping Up HIV Testing in the Era of COVID-19 : PSI. Available from: https://www.psi.org/2020/05/ramping-uphiv-testing-in-the-era-of-covid-19/?utm_source=rss&utm_medium=rss&utm_campaign=ramping-up-hiv-testing-in-theera-of-covid-19 [Last accessed on 2020 Jul 29]
- [Google Scholar]
- Experiences of health care providers with integrated HIV and reproductive health services in Kenya: A qualitative study. BMC Health Serv Res. 2013;13:18.
- [CrossRef] [PubMed] [Google Scholar]
- Youth accessing reproductive health services in Malawi: Drivers, barriers, and suggestions from the perspectives of youth and parents. Reprod Health. 2018;15:108.
- [CrossRef] [PubMed] [Google Scholar]
- Are one-stop shops acceptable? Community perspectives on one-stop shop models of sexual health service provision in the UK. Sex Transm Infect. 2008;84:395-9.
- [CrossRef] [PubMed] [Google Scholar]
- Zimbabwe Human Development Report 2017: Climate Change and Human Development: Towards Building a Climate Resilient Nation. In: Harare. Zimbabwe: UNDP; 2017.
- [Google Scholar]
- UNAIDS Country: Zimbabwe. 2016. Available from: https://www.unaids.org/en/regionscountries/countries/zimbabwe [Last accessed on 2018 Apr 17]
- [Google Scholar]
- Ministry of Health and Child Welfare, National Tuberculosis Program-Strategic Plan (2017-2020) Harare, Zimbabwe: Zimbabwe Ministry of Health and Child Care; 2018.
- [Google Scholar]
- Population of Harare in 2020 2021-Statistics. Available from: https://all-populations.com/en/zw/population-of-harare.html [Last accessed on 2021 Oct 05]
- [Google Scholar]
- City of Harare: Council Health Facilities. Available from: https://www.hararecity.co.zw/services/about/council-health-facilities [Last accessed on 2021 Oct 05]
- [Google Scholar]
- Bulawayo City Council. Available from: https://www.citybyo.co.zw [Last accessed on 2021 Oct 06]
- [Google Scholar]
- Integrating pre-exposure prophylaxis delivery in routine family planning clinics: A feasibility programmatic evaluation in Kenya. PLoS Med. 2019;16:e1002885.
- [CrossRef] [PubMed] [Google Scholar]
- Assessing Provider-, Clinic-, and structural-level barriers and recommendations to pre-exposure prophylaxis (PrEP) uptake: A qualitative investigation among women experiencing intimate partner violence, intimate partner violence service providers, and healthcare providers. AIDS Behav. 2021;25:3425-36.
- [CrossRef] [PubMed] [Google Scholar]
- Predictors of HIV-related risk perception and PrEP acceptability among young adult female family planning patients. AIDS Care. 2017;29:751-8.
- [CrossRef] [PubMed] [Google Scholar]
- HIV risk' perceptions among adolescents attending family planning clinics: An integrated perspective. AIDS Care. 2013;25:20-7.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of sexually transmitted disease/HIV risk among young African Americans: Comparison of self-perceived and epidemiological risks utilizing ecodevelopmental theory. HIV AIDS (Auckl). 2019;11:31-44.
- [CrossRef] [PubMed] [Google Scholar]
- African-American adolescents residing in high-risk urban environments do use condoms: Correlates and predictors of condom use among adolescents in public housing developments. Pediatrics. 1996;98:269-78.
- [CrossRef] [PubMed] [Google Scholar]
- Cross-cultural comparison of US and Nigerian adolescents' HIV-related knowledge, attitudes, and risk behaviour: Implications for risk reduction interventions. AIDS Care. 1995;7:449-62.
- [CrossRef] [PubMed] [Google Scholar]
- How can we help adolescent girls avoid HIV infection? Netw Res Triangle Park N C. 1993;13:7.
- [Google Scholar]
- The ECHO Study Press Releases: Wits RHI. Available from: https://www.wrhi.ac.za/expertise/detail/the-echo-study [Last accessed on 2021 Feb 04]
- [Google Scholar]
- Sexual risk and perception of risk for HIV infection among multiethnic family-planning clients. Am J Prev Med. 1993;9:92-5.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of sexually transmitted disease/HIV risk among young African Americans: Comparison of self-perceived and epidemiological risks utilizing ecodevelopmental theory. HIV/AIDS (Auckl). 2019;11:31-44.
- [CrossRef] [PubMed] [Google Scholar]
- HIV risk perceptions among adolescents attending family planning clinics: An integrated perspective. AIDS Care. 2013;25:20-7.
- [CrossRef] [PubMed] [Google Scholar]
- Family Health International (FHI 360): Family Planning and HIV Integration: Approaching the Tipping Point. 2022. Available from: https://www.fhi360.org/sites/default/files/media/documents/FPHIVoverview1.pdf
- [Google Scholar]
- Male partner testing and sexual behaviour following provision of multiple HIV self-tests to Kenyan women at higher risk of HIV infection in a cluster randomized trial. J Int AIDS Soc 2020(Suppl 2):e25515.
- [CrossRef] [PubMed] [Google Scholar]
- Acceptability and outcomes of distributing HIV self-tests for male partner testing in Kenyan maternal and child health and family planning clinics. AIDS. 2019;33:1369-78.
- [CrossRef] [PubMed] [Google Scholar]
- Promoting partner testing and couples testing through secondary distribution of HIV self-tests: A randomized clinical trial. PLoS Med. 2016;13:e1002166.
- [CrossRef] [PubMed] [Google Scholar]
- Integration of STI and HIV prevention, care, and treatment into family planning services: A review of the literature. Stud Fam Plann. 2009;40:17-8.
- [CrossRef] [PubMed] [Google Scholar]






